
LEO Foundation Center for Cutaneous Drug Delivery
The aim of the LEO Foundation Center for Cutaneous Drug Delivery is to contribute to an integrating knowledge on skin as a barrier for drug absorption, on the one hand, and the properties of drugs and excipients in specific drug delivery systems, on the other.
The skin is an organ that often requires drug treatment to alleviate acute and chronic skin diseases. Furthermore, it is easily accessible for application of drugs, and thus also a site for administration of drugs aiming for systemic effects. However, the skin remains a formidable barrier and the delivery of drugs into and across the skin barrier is highly dependent on a number of factors in a complex way.
Considering this, the aim of the LEO Foundation Center for Cutaneous Drug Delivery is to contribute to an integrating knowledge on skin as a barrier for drug absorption, on the one hand, and the properties of drugs and excipients in specific drug delivery systems, on the other.
Through this, we aim to establish an internationally competitive research environment for cutaneous drug delivery research within a 10-year period, based on a predominantly physicochemical approach to develop methods for the rational development of novel and improved drug delivery systems for cutaneous and transcutaneous delivery of both small and large molecules. In doing so, investigations of "on-site" properties of the drug delivery system, taking into account effects in both healthy and impaired skin, play a central role.
With the main outset to build on a physicochemical basis, the Center aims to be clearly visible within the field of cutaneous drug delivery, and differentiated from approaches based on a more empirical development of delivery systems and on clinical work. This approach also allows the Center to be a pivotal point for collaborations with other research fields, ultimately coming together to tackle fundamental questions related to the delivery of drugs to and across the skin.
The LEO Foundation Center for Cutaneous Drug Delivery is made possible by a 10-year grant from the LEO Foundation.
Vision
To become an international focal point for harnessing the potential of physical chemistry for the rational design of cutaneous drug delivery systems that can spawn the next generation of minimally invasive, targeted, and effective dermatological therapies.
Mission
We will achieve our vision by:
- integrating cutting-edge knowledge and approaches from physical chemistry with biology, pharmaceutics, and nanotechnology
- harnessing national and international collaborations among academia, industry, and the clinic
- training the next generation of scientists in the field of cutaneous drug delivery.
Strategic Objectives
- To understand the processes governing transport of molecules across the skin barrier and its biological consequences by integrating state-of-the-art physicochemical methods and novel materials with biology and pharmaceutics.
- To undertake the rational design of systems for effective drug delivery through the skin barrier.
- To inspire industrial and clinical partners to explore pathways for translational therapies based on cutaneous drug delivery approaches developed within the Center.
- To provide a high-quality, holistic training platform on cutaneous drug delivery, encompassing analytical methods, drug delivery, and biological effects.
- To harness the infrastructure and physical-chemistry expertise in the Øresund region by forging links with MAX IV/ESS.
LFCCDD is embedded fully within the Department of Pharmacy, University of Copenhagen, and rests on three pillars:
(i) novel analytical tools
(ii) novel drug delivery systems
and (iii) novel biological models
Each of these is led by a tenure-track Assistant/Associate Professor (TT), supported by an experienced Professor (PI) for mentorship and supervision support. Each TT leads an emerging research group of PhD students and Postdocs.
The research in the three pillars is coordinated through project design and shared supervision for maximum synergies and shared infrastructure handling. The overall Center activities is overseen by a Center Director. In total, the Center hosts about 20 group members.
Martin Malmsten, Research Director & Principle Investigator
His current research interests include antimicrobial nanomaterials and nanomaterials for drug delivery, amphiphilic peptide therapeutics, and cutaneous drug delivery. In addition to his academic work, Malmsten is active within research translation, being co-inventor on 16 patent and patent application families, as well as co-founder for three start-ups. Malmsten has received numerous research awards, including the ACS Langmuir Lecture Award and the Nordblad-Ekstrand Medal. He is elected Fellow of the Royal Swedish Academy of Sciences (Nobel Prize awarding academy), the Royal Swedish Academy of Engineering Sciences (where he was Chairman of Chemical Engineering 2018-20), and the Danish Academy of Technical Sciences. In addition, Malmsten is Fellow of the Royal Physiographic Society, Fellow of the Royal Chemical Society, and guest professor at Lund University. He is also Editor-in-Chief of the Journal of Colloid and Interface Science, one of the leading journals within colloid and interface science, with an impact factor of 9.96 and over 10,000 manuscript submissions per year. |
Thomas Rades, Principle Investigator – Novel Drug Delivery Systems
|
Hanne Mørck Nielsen, Principle Investigator – Novel Biological Models
|
Jesper Østergaard, Principle Investigator – Novel Analytical Technologies
|
|
Prof. Richard Guy, Department of Pharmacy and Pharmacology, University of Bath, UK. Richard Guy received an M.A. in Chemistry from Oxford University, and his Ph.D. in Pharmaceutical Chemistry from the University of London. He has held academic posts at the University of California, San Francisco, the University of Geneva and, since 2004, the University of Bath as Professor of Pharmaceutical Sciences. In 2013, Dr. Guy received the Controlled Release Society's Founders Award and, in 2016, he was awarded the degree of Doctor of Science from Oxford University. Dr. Guy's research focuses on skin barrier function characterization and the prediction, assessment and optimization of topical drug bioavailability. He has published 350+ peer-reviewed articles and over 70 book chapters. He has co-authored one book and co-edited 7 others. He is also co-inventor of 12 patents. Within the Scientific Advisory Board, Prof. Guy will provide expertise in dermal drug delivery. |
| Prof. Frank Caruso, Chemical and Biomolecular Engineering, University of Melbourne, Australia. Professor Caruso is ARC Australian Laureate Fellow at The University of Melbourne, Australia, and also Deputy Director, ARC Centre of Excellence on Convergent Bio-Nano Science and Technology. He is also leader of the Nanostructured Interfaces and Materials Science (NIMS) Group. He received his PhD degree in 1994 from the University of Melbourne, and then moved to the CSIRO Division of Chemicals and Polymers in Melbourne. He was an Alexander von Humboldt Research Fellow and then group leader at the Max Planck Institute of Colloids and Interfaces (Berlin, Germany) from 1997 until 2002. His research interests focus on developing advanced nano- and biomaterials for biotechnology and medicine. He has published over 300 peer-reviewed papers and is on the ISI most highly cited list, ranking in the top 20 worldwide, in materials science in 2011. Within the Scientific Advisory Board, Prof. Caruso will provide expertise in nanotechnology. |
| Prof. Lise Arleth, Neutron and X-Ray Science Group, Niels Bohr Institute, University of Copenhagen, Denmark. Professor Arleth works within biophysics and physical chemistry with the main focus on structural investigations of macromolecules and their aggregates in solution. The research involves self-organizing systems such as microemulsions, surfactants and polymers in solution, as well as more complex biological and/or pharmaceutically relevant systems, such as proteins/enzymes and their interactions with lipids and surfactants. Her main experimental technique is small-angle scattering (X-rays and neutrons), in which she is an acclaimed international expert. Professor Arleth has been, and still is, instrumental for the engagement of the University of Copenhagen in MAX-Lab IV and ESS in Lund. She is the author over more than 75 peer-reviewed papers, and is also associated with the Center of Synthetic Biology. Within the Scientific Advisory Board, Prof. Arleth will provide expertise in X-ray and neutron-based analytical tools for investigation of soft matter. |
Research within LFCCDD is built on integrating physicochemical concepts and methodologies with novel opportunities in nanotechnology, advanced analytical methodologies, and biological models. It rests on three pillars:
1. Novel analytical tools
2. Novel drug delivery systems
3. Novel biological models
Each pillar is led by a tenure-track Assistant/Associate Professor (TT). The research pillars are pooled to address the over-arching research ambition to overcome the skin barrier by understanding its physicochemical properties and using this understanding for rationally designing cutaneous drug delivery systems.
As such, the research focus: 1. explores a more focused set of important physicochemical mechanisms; 2. prioritizes the mechanistic foundation for designing drug delivery systems; and 3. strengthens synergies between the research pillars.
The research capitalizes on recent developments within materials science and on understanding of the structure and dynamics of novel drug delivery systems, as well as state-of-the-art methodologies for their investigation.
Based on this, we aim to:
- Understand the processes governing transport of molecules across the skin barrier and its biological consequences.
- Undertake the rational design of systems for effective drug delivery through the skin barrier.
- Harness the infrastructure and physical-chemistry expertise in the Øresund region by forging links links with large-scale facilities such as MAX IV/ESS.
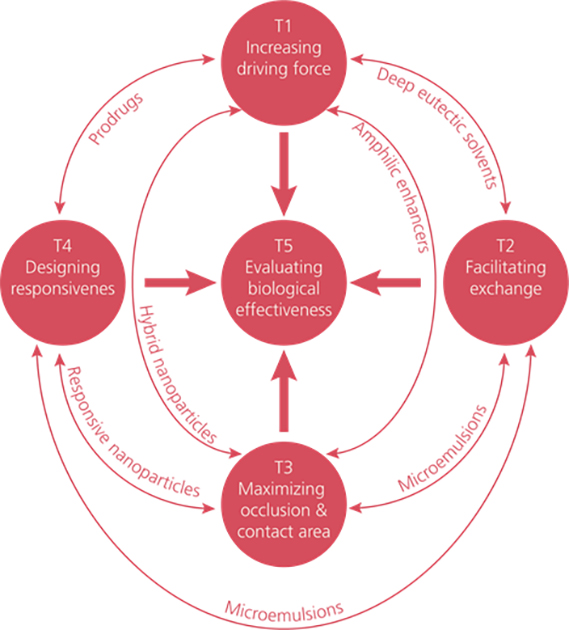
Currently, particular focus lies on a combined efforts for exploring four different physicochemical processes to overcome the skin barrier (see illustration below), and to elucidate the biological effects of overcoming the barrier.
Permeation through increased driving force
-Multiple prodrugs for maximized chemical potential
-Stabilization of supersaturated states
Permeation through facilitated exchange
-Deep eutectic solvents
-Microemulsions
Permeation through maximized occlusion and surface area
Defect-free film formation
Permeation through designed responsiveness
Infection- and inflammation triggered systems
The four different physicochemical processes that are essential in order to overcome the skin barrier
Contact
Research Director and
Principle Investigator
Professor
Martin Malmsten
+45 31 49 92 03
martin.malmsten@sund.ku.dk
Associate Professor
Andrea Heinz
+ 45 35 33 77 83
andrea.heinz@sund.ku.dk
Associate Professor
Mariena van der Plas
+45 35 33 27 16
mariena.van_der_plas
@sund.ku.dk
Assistant Professor
Stine Rønholt
+45 35 33 73 13
stine.roenholt@sund.ku.dk

 Martin Malmsten is professor at the University of Copenhagen and founding Director of the LEO Foundation Center for Cutaneous Drug Delivery. Previously, he was professor and Head of Department at Uppsala University, and before that CEO of YKI, Institute for Surface Chemistry AB.
Martin Malmsten is professor at the University of Copenhagen and founding Director of the LEO Foundation Center for Cutaneous Drug Delivery. Previously, he was professor and Head of Department at Uppsala University, and before that CEO of YKI, Institute for Surface Chemistry AB.  Professor in Pharmaceutical Design and Drug Delivery at the Department of Pharmacy, University of Copenhagen.
Professor in Pharmaceutical Design and Drug Delivery at the Department of Pharmacy, University of Copenhagen. Professor in Biopharmaceuticals and Drug Design, and Research Chair of the Section of Biologics at Department of Pharmacy, University of Copenhagen.
Professor in Biopharmaceuticals and Drug Design, and Research Chair of the Section of Biologics at Department of Pharmacy, University of Copenhagen. Associate Professor at Department of Pharmacy, University of Copenhagen, and leader of the Pharmaceutical and Physical Chemistry group.
Associate Professor at Department of Pharmacy, University of Copenhagen, and leader of the Pharmaceutical and Physical Chemistry group.